Zydus Pharmaceuticals is recalling approximately 2,454 packs of Acyclovir Sodium Injection (500mg/10mL and 1000mg/20mL) because crystals have formed inside the finished product. Acyclovir is an antiviral drug used to treat infections caused by herpes viruses, and these specific lots are administered by intravenous infusion. The defect was discovered following customer complaints of crystallization in the vials. You should check your medication for the specific lot numbers and expiration dates listed below and consult your doctor if you have this product.
Injecting a product containing crystals can cause serious health problems, such as inflammation or blockages in the blood vessels, which could lead to organ damage or other life-threatening complications.
Drug recall baseline remedy

Acyclovir Sodium Injection, 500 mg/10mL vial
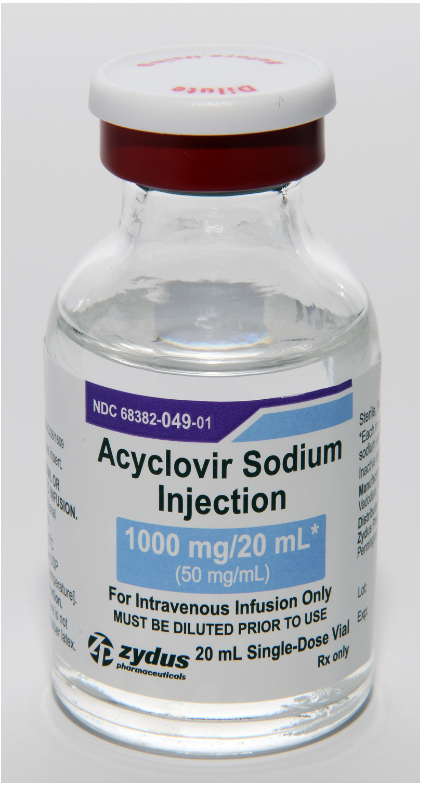
Acyclovir Sodium Injection, 1000 mg/20mL vial
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.