Trividia Health is recalling approximately 18.2 million owner's booklets and instructions for several TRUE METRIX blood glucose monitoring systems. The instructions do not adequately explain that an 'E-5' error code on the meter may indicate a life-threateningly high blood sugar level. The manuals also fail to instruct users to seek immediate medical attention when this error occurs. These systems were sold at major retailers including Walgreens, CVS, Walmart, Rite Aid, and Kroger.
If a user has very high blood sugar and receives an E-5 error code, they may not realize they are in a medical emergency. This can lead to a dangerous delay in seeking treatment, potentially resulting in serious health complications or death from untreated high glucose.
You have 2 options:
Includes various store-branded meters such as Walgreens, CVS, Leader, GNP, Meijer, and Rite Aid.
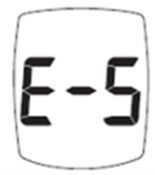
"Image of E-5 error code on screen"
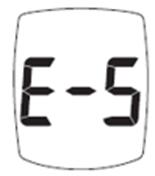
"Image of E-5 error code on screen"
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.