Novartis Consumer Health is recalling approximately 2.3 million units of Triaminic Syrups and Theraflu Warming Relief Syrups (acetaminophen and diphenhydramine) used for coughs, colds, and fevers. The child-resistant caps on these bottles can fail, allowing children to remove them even with the tamper-evident seal in place. The company has received 12 reports of children unscrewing the caps, including four instances of ingestion, with one child requiring medical attention. These products were sold at food, drug, mass merchandise, and club stores nationwide between May 2010 and December 2011 for about $5. Consumers should immediately stop using the recalled syrups and contact Novartis for a full refund.
The faulty caps allow children to easily access medications containing acetaminophen and diphenhydramine, posing a serious risk of accidental ingestion and poisoning. Four children are reported to have ingested the product after opening the locked caps.
Consumers should immediately stop using the recalled product and contact Novartis for instructions on how to return the product for a full refund.
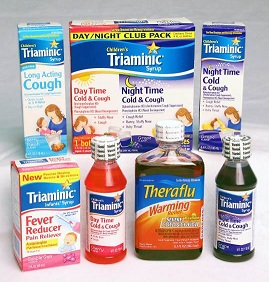
Triaminic Syrups and Theraflu Warming Liquids
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response