Teva Pharmaceuticals USA Inc. has voluntarily recalled 7,047 units of Fentanyl Buccal Tablets (fentanyl) distributed by Mayne Pharma. These prescription tablets, used for severe pain management, were distributed with incorrect or missing package inserts. Without the proper documentation, patients and healthcare providers may lack critical safety information, usage instructions, and warnings necessary for the safe administration of this potent medication. No injuries or adverse incidents have been reported to date.
The absence of a correct package insert means users may not have access to essential safety warnings and dosage instructions, increasing the risk of improper use or serious adverse health consequences. This is particularly critical given the high potency of fentanyl-based medications.
You have 2 options:
Distributed by Mayne Pharma
Distributed by Mayne Pharma
Distributed by Mayne Pharma
Distributed by Mayne Pharma
Distributed by Mayne Pharma

Image 1 -Carton labeling, Fentanyl Buccal Tablets, 100 mcg
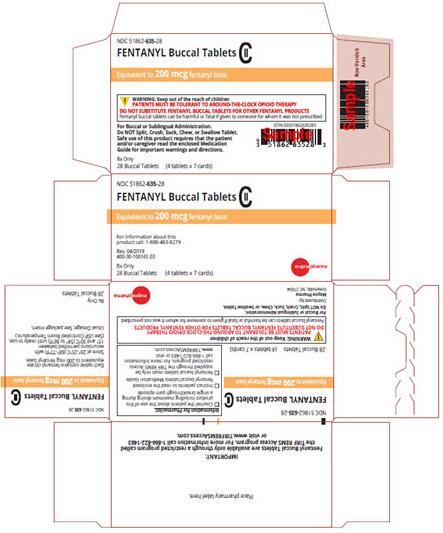
Image 2 - Carton labeling, Fentanyl Buccal Tablets, 200 mcg
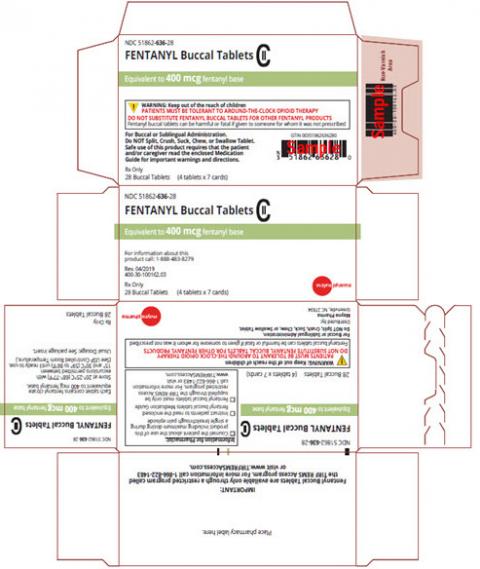
Image 3 – Carton labeling, Fentanyl Buccal Tablets, 400 mcg

Image 4 - Carton labeling, Fentanyl Buccal Tablets, 600 mcg
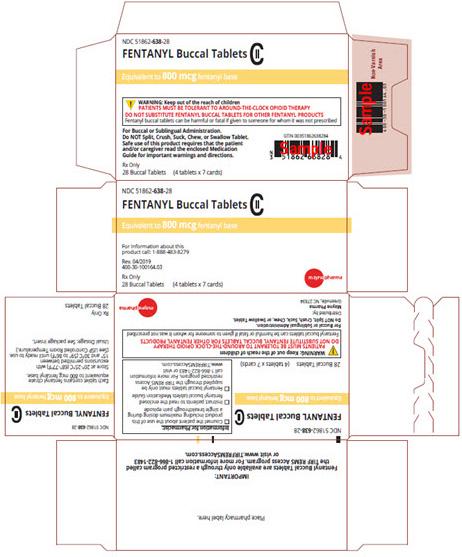
Image 5 - Carton labeling, Fentanyl Buccal Tablets, 800 mcg
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.