Jichehui Electronics Co. Ltd., doing business as TecFlox, is recalling approximately 900 bottles of TecFlox Minoxidil Hair and Beard Growth Serum. The serum contains minoxidil, a substance that requires child-resistant packaging under the Poison Prevention Packaging Act, which these bottles lack. No incidents or injuries have been reported to date. This topical solution was sold exclusively on Amazon.com in October 2025.
The bottles are not child-resistant, posing a risk of serious injury or death from poisoning if a young child accidentally swallows the contents. This violates federal safety standards meant to protect children from hazardous substances.
Consumers should immediately secure the recalled serum bottles out of sight and reach of children and contact TecFlox to receive free replacement serum bottles, including shipping. Only bottles with serum remaining will be replaced. Consumers will be asked to dispose of the bottles' contents and email a photo of the bottles in the trash, showing the manufacture date to TecFloxrecall@outlook.com
Sold in packs of four 60mL bottles. The manufacture date is located on the underside of the bottle.
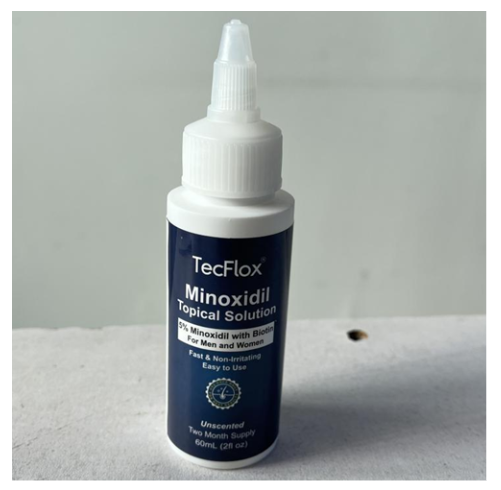
Recalled TecFlox Topical Solution - hair and beard growth serum bottle

The manufacture date "MFD 2025.8.25" is printed on the underside of the recalled TecFlox hair and beard growth bottles

Recalled TecFlox Topical Solution - hair and beard growth serum packaging
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response