Stratford Care USA, Inc. is recalling 5,254 bottles of Omega-V3 and Skin+Coat pet softgels because they contain elevated levels of Vitamin A. Feeding these supplements to pets can lead to serious health issues including Vitamin A toxicity. The affected products were distributed to specialty retailers and veterinary clinics across the United States. While no incidents or injuries have been reported, consumers should immediately stop using these supplements and check their packaging for specific lot numbers.
Excessive Vitamin A intake can cause acute or chronic toxicity in dogs and cats, leading to symptoms like malaise, skin peeling, joint pain, and in severe cases, permanent organ damage or death. The recalled softgels contain levels of Vitamin A that significantly exceed nutritional requirements for small and medium pets.
You have 2 options:
Distributed quantity: 5,254 bottles of lot #31133 and 6,198 bottles of lot #30837.

Multi brand labels for Omega-3 Soft Gel Supplements for cats and dogs, see table above

Multi brand labels for Omega-3 Soft Gel Supplements for cats and dogs, see table above

Multi brand labels for Omega-3 Soft Gel Supplements for cats and dogs, see table above

Multi brand labels for Omega-3 Soft Gel Supplements for cats and dogs, see table above

Multi brand labels for Omega-3 Soft Gel Supplements for cats and dogs, see table above

Multi brand labels for Omega-3 Soft Gel Supplements for cats and dogs, see table above
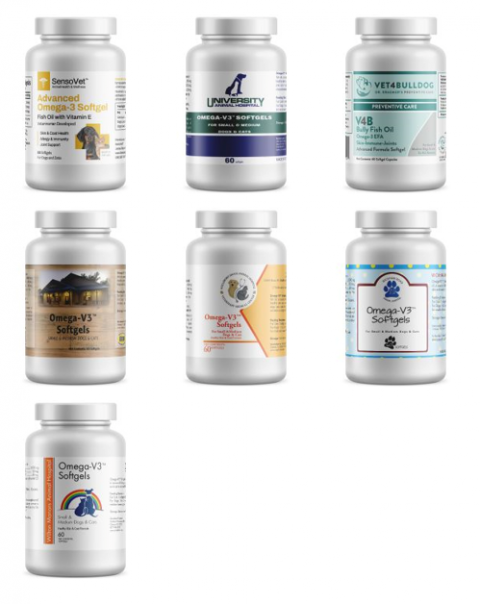
Multi brand labels for Omega-3 Soft Gel Supplements for cats and dogs, see table above
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response