Southwind Foods LLC is recalling 11,081 cases across all products and brands of frozen raw and cooked shrimp sold under the Sand Bar, Great American, First Street, Best Yet, and Arctic Shores brands. The recall was initiated because the products were manufactured under insanitary conditions where they may have been contaminated with Cesium-137 (Cs-137). The affected shrimp was distributed in Alabama, Arizona, California, Massachusetts, Minnesota, Pennsylvania, Utah, Virginia, and Washington state.
Cesium-137 is a radioactive isotope. Consuming food contaminated with this substance can increase the risk of cancer. While no illnesses have been reported, the manufacturing conditions did not meet required safety standards.
You have 2 options:
Recall #: H-0603-2025
Recall #: H-0604-2025

FROZEN RAW SHRIMP 31/40 SAND BAR 2LB BAG

SHRIMP 26/30 CPDTO WHITE BEST YET

FROZEN COOKED SHRIMP 31/40 BEST YET 1LB BAG

FROZEN COOKED SHRIMP 31/40 BEST YET 1 LB BAG

FROZEN COOKED SHRIMP 41/50 BESY YET 1LB BAG

FROZEN LARGE COOKED SHRIMP 31/40 ARTIC SHORES 1LB BAG

FROZEN SMALL COOKED SHRIMP 61/70 ARTIC SHORES 1LB BAG

FROZEN SMALL COOKED SHRIMP 91/120 CPUDT/OFF WHITE ARCTIC SHORES 12OZ BAG

FROZEN COOKED SALAD SHRIMP 150-200 ARCTIC SHORES 6OZ BAG

FROZEN RAW SHRIMP 16/20 GREAT AMERICAN 2LB BAG

FROZEN RAW SHRIMP 16/20 FIRST STREET 2LB BAG

FROZEN RAW SHRIMP 71/90 GREAT AMERICAN 2LB BAG

FROZEN COOKED SHRIMP MEAT GREAT AMERICAN 1LB BAG

FROZEN COOKED SHRIMP 41/60 GREAT AMERICAN 2LB BAG
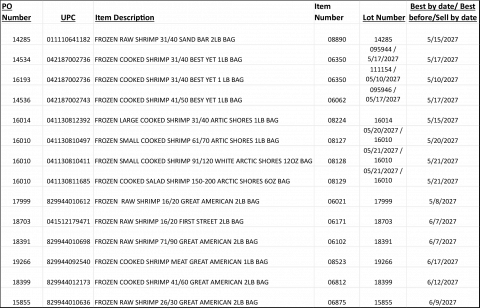
Product List
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release (1) · FDA Press Release (2) · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.