SleepNet Corporation is recalling 45,972 CPAP and nasal masks, including the Mojo, Mojo 2, iQ 2, and Phantom 2 models, to update safety warnings and contraindications. These masks contain magnets that can interfere with certain implanted medical devices, such as pacemakers or brain shunts. This voluntary recall was initiated to ensure users are aware of the risks when using these masks near metallic implants or devices sensitive to magnetic fields.
The magnets used in these masks can potentially cause implanted medical devices to malfunction or shift if they are within close proximity. This magnetic interference could lead to serious health complications or the failure of life-saving equipment like implanted defibrillators or neurostimulators.
Safety labeling update

Image of Phantom 2 Nasal Mask
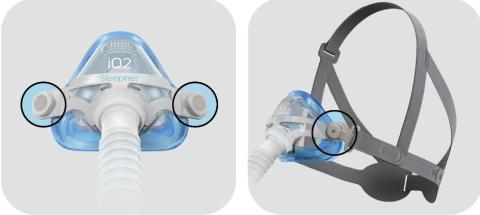
Image of iQ 2 Nasal Mask
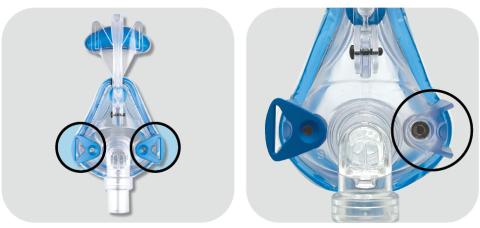
Image of Mojo Full Face Vented Mask and Mojo Full Face Non Vented Mask

Image of Mojo 2 Full Face Vented Mask, Mojo 2 Full Face Non Vented Mask, and Mojo 2 Full Face AAV Non Vented Mask
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.