Sanvall Enterprises is recalling approximately 5,400 units of Rapid Alivio Pain Relieving Roll-On (methyl salicylate). The product is being recalled because its packaging is not child-resistant as required by federal law for substances containing methyl salicylate. This failure to meet safety requirements creates a danger of poisoning if a young child accesses and swallows the liquid. No injuries have been reported, and affected bottles were sold at Walmart and Navarro Discount Pharmacy in South Florida between August 2016 and June 2019. Consumers should contact the company for a full refund.
The bottle lacks child-resistant packaging, making it easy for young children to open and swallow the contents. If ingested, the active ingredient methyl salicylate can cause serious illness or fatal poisoning.
Consumers should immediately store the pain relieving roll-on in a safe location out of reach of children and contact Sanvall Enterprises for a full refund.
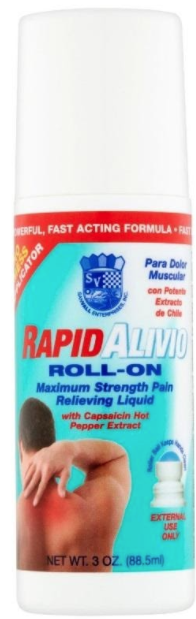
Recalled Sanvall Rapid Alivio Pain Relieving Roll-On – 3 fl. oz (88.5 mL)

Recalled Sanvall Rapid Alivio Pain Relieving Roll-On – label

UPC code 605100014225 is printed on the side of the label.
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response