Zhongshan Biankang Medical Equipment Co., Ltd. (dba Sangohe) is recalling about 26,200 Sangohe-branded adult portable bed rails sold on Amazon.com and Walmart.com between August 2023 and October 2025. These bed rails violate mandatory safety standards because users can become trapped within the rail or between the rail and the mattress, which can lead to serious injury or death. To receive a full refund, consumers should stop using the bed rails and destroy them by cutting the foam padding and marking them as "RECALLED" before emailing proof to the manufacturer.
Users can become trapped in the bed rail or wedged between the rail and the mattress, posing a serious risk of entrapment and death by suffocation (asphyxiation). Additionally, the products are missing required hazard warning labels.
Consumers should immediately stop using the recalled bed rails and contact Sangohe for a full refund. Consumers should destroy the bed rails by cutting the handrails' foam padding and writing "RECALLED" on the upper and lower rails with permanent marker, take a photo of the destroyed rails and email the photo to SGHproductrecall@163.com.
Model number is printed on a label located on the frame, either on the bottom foot tube or on the horizontal cross tube. 'Sangohe' is printed on the packaging.

Recalled Sangohe Bed Rail - front view

Recalled Sangohe Bed Rail - side view

Recalled Sangohe Bed Rail - top view

Model number "KDB504A01FT" is printed on a label located on the frame, either on the bottom foot tube or on the horizontal cross tube of the recalled Sangohe bed rail

Model number "KDB504A01FT" is printed on a label located on the frame, either on the bottom foot tube or on the horizontal cross tube of the recalled Sangohe bed rail
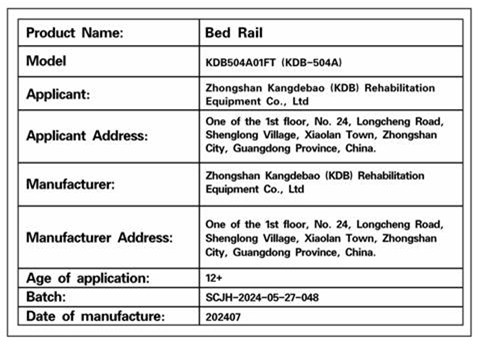
Label showing model number "KDB504A01FT"
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response