Sandoz Inc. is recalling approximately 156,750 units of Aprepitant (125 mg) capsules and fougera Lidocaine and Prilocaine (2.5%/2.5%) cream. These prescription medications do not have child-resistant packaging as required by the Poison Prevention Packaging Act. No injuries have been reported, but consumers should immediately secure these medicines away from children and contact Sandoz for a free child-resistant storage pouch.
The current packaging is not child-resistant, which means children can easily open the containers and swallow the contents. This poses a significant risk of accidental poisoning if the medication is ingested by a child.
Consumers should immediately secure the medications out of the sight and reach of children and contact Sandoz for a free child resistant pouch to store the products. Once the medication is secured, consumers can continue to use the medication as directed.
Sold as a carton containing one blister card of 6 capsules.
Sold in cartons containing 5 tubes, with or without 12 dressings.

Recalled Aprepitant Capsules 125 mg – NDC 0781-2323-68 – Carton

Recalled Aprepitant Capsules 125 mg – NDC 0781-2323-06 – Bliste

Recalled Lidocaine and Prilocaine Cream - 5 gram tube - NDC 0168-0357-05
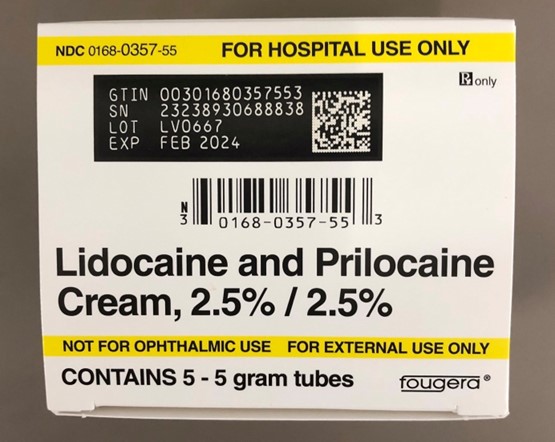
Recalled Lidocaine and Prilocaine Cream - Carton of 5 – 5 gram tubes - NDC 0168-0357-55

Recalled Lidocaine and Prilocaine Cream - Carton of 5 – 5 gram tubes with 12 dressings - NDC 0168-0357-56
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response