Novartis and Sandoz are recalling approximately 470,000 units of various prescription drug blister packages because the packaging is not child-resistant as required by law. The medications affected include several common drugs like Zofran ODT (ondansetron), Entresto (sacubitril/valsartan), and various generic tablets such as Donepezil, Haloperidol, and Risperidone. These medications were sold at clinics and pharmacies nationwide from September 2016 through June 2018. Consumers should immediately secure the affected medications and contact the manufacturers for further instructions.
The packaging fails to meet child-resistant closure requirements, posing a serious poisoning risk if the medication is accessed and swallowed by a child. There has been one report of a child ingesting haloperidol from one of these blister packs.
Consumers should immediately secure the blister cards to keep them out of the sight and reach of children and contact Novartis or Sandoz for further instructions. Novartis and Sandoz advise that consumers should continue to use the medication as directed once the blister packages are secured.

Donepezil ODT 10 mg
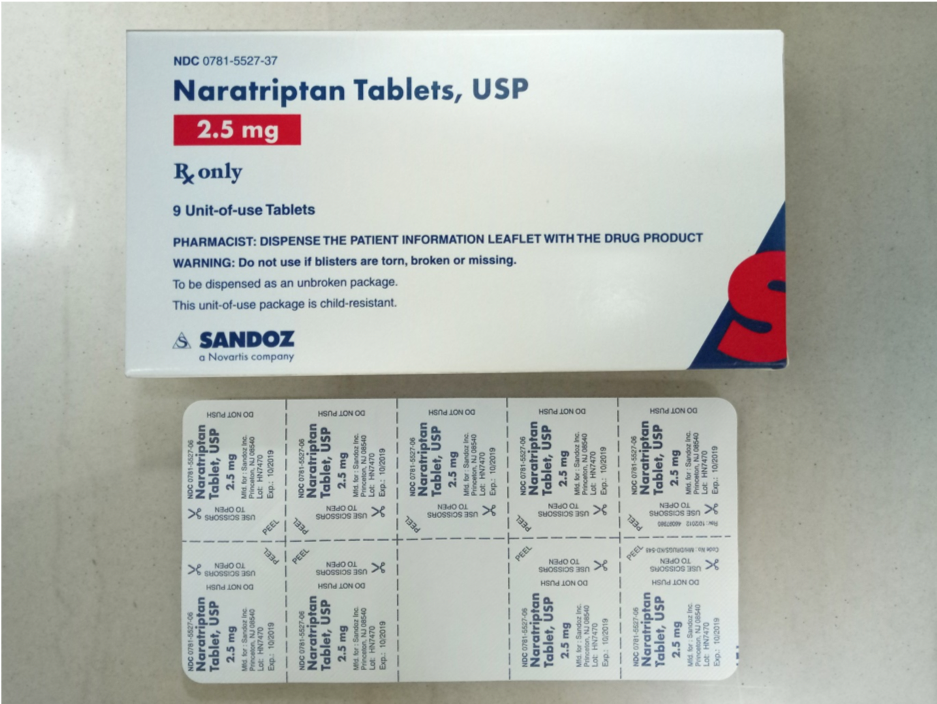
Naratriptan 2.5 mg tablets
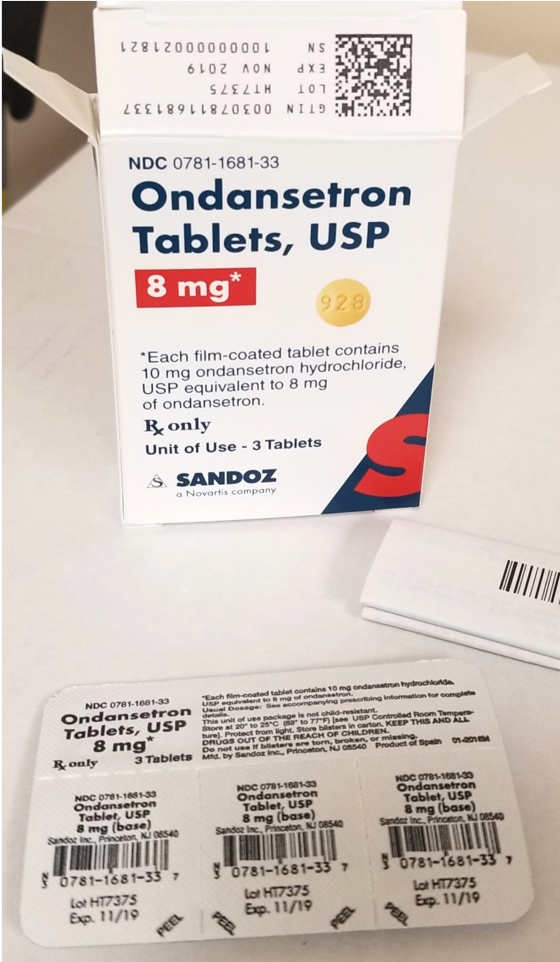
Ondansetron Tablets 8 mg.

Risperidone ODT 2 mg.
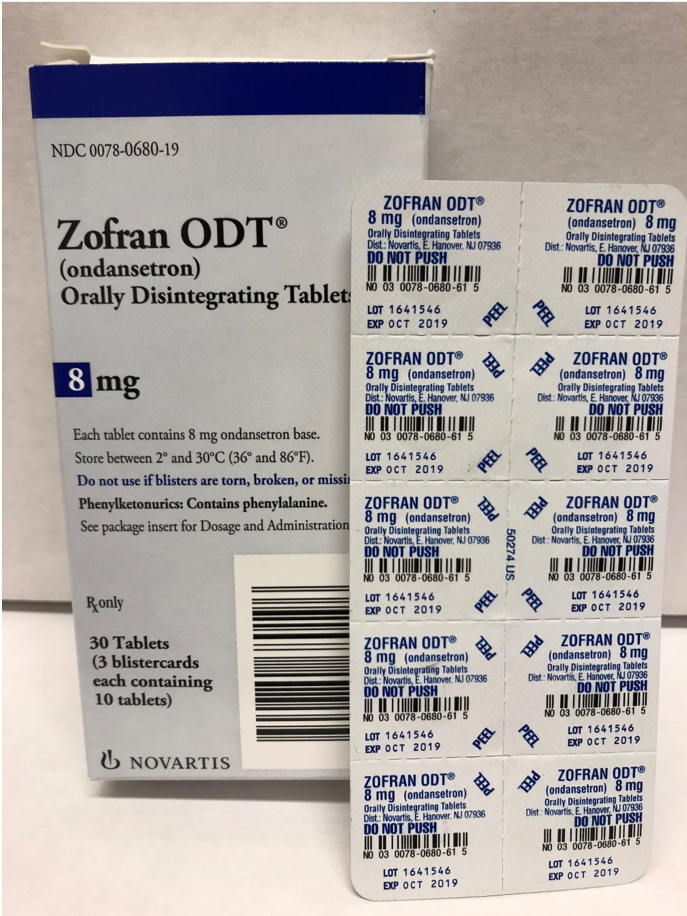
Zofran® ODT 4 mg.
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response