Savaria Corporation has recalled approximately 19,500 residential elevators branded under the Savaria and Garaventa names, including models Eclipse, Infinity, Kwiklift, MR, and HR. These elevators were manufactured between 1999 and 2021 and sold for residential use across the United States. The recall addresses a hazardous gap between the exterior landing door and the interior elevator car door that can entrap children, posing a serious risk of injury or death. Consumers should keep children away from the elevators and contact Savaria for free space guards and installation assistance.
Young children can become entrapped in the space between the exterior landing (hoistway) door and the interior elevator car door or gate. If the elevator is called to another floor while a child is in this gap, the child can be crushed or pinned, leading to serious injury or death.
Consumers should keep unsupervised young children away from the recalled residential elevators and contact the manufacturers for instructions on how to measure for space guards to correct any hazardous gap. Space guards will be provided free of charge and assistance with space guard installation will be provided on request.
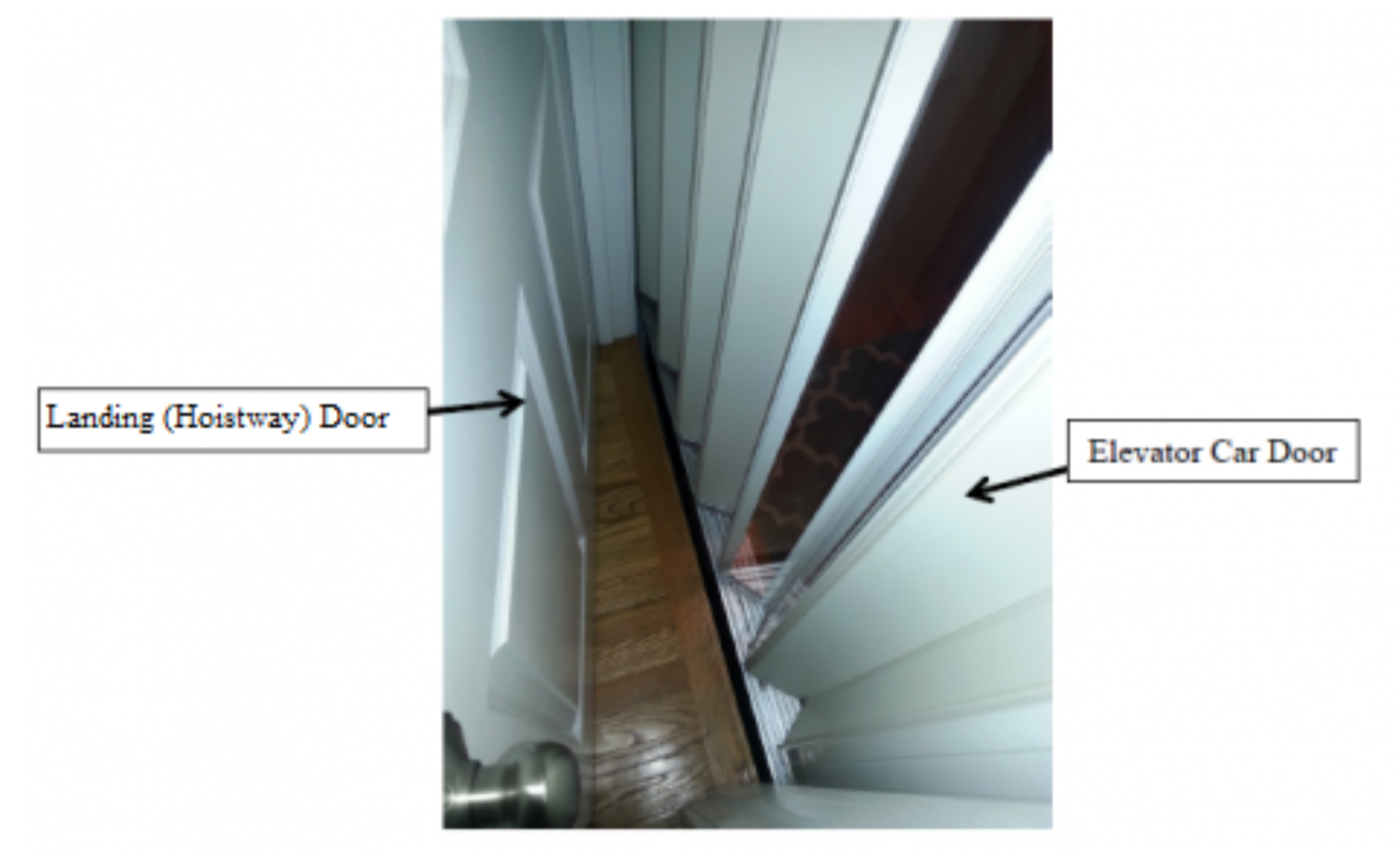
Residential Elevator with Space Between the Exterior Landing (Hoistway) Door and Interior Elevator Car (Accordion) Door. A young child can become entrapped if there is a hazardous gap between the doors.
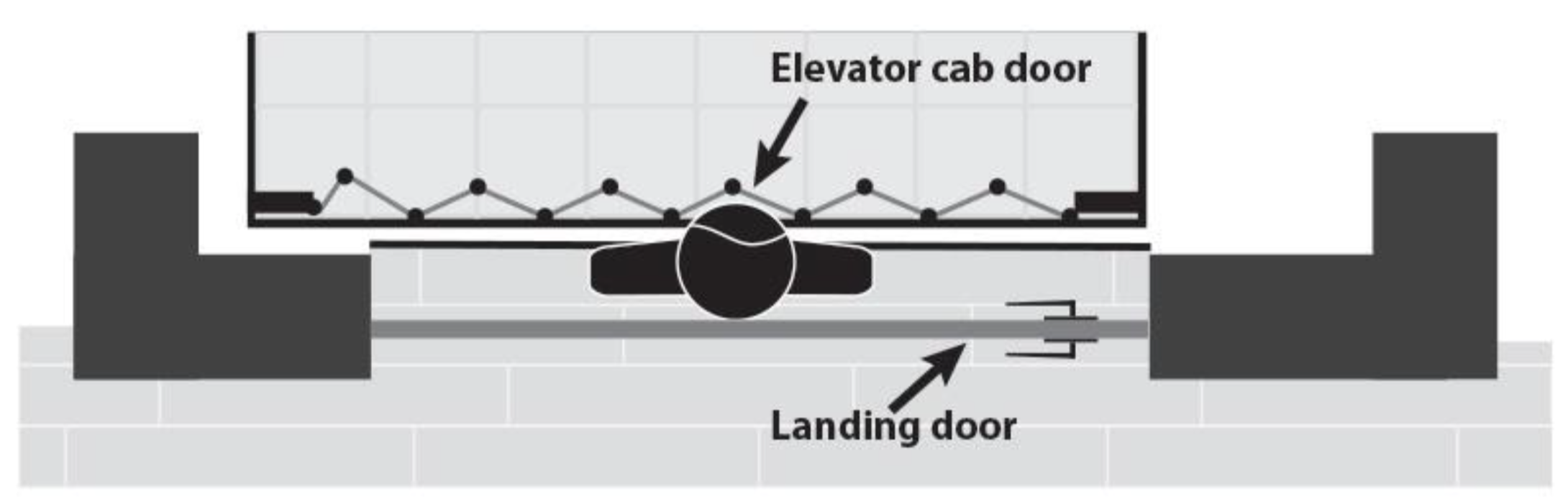
Scenario depicting a child trapped between an exterior landing (hoistway) door and an interior elevator car door due to a hazardous gap. The exterior door locks the young child in the space between the doors when the elevator is called to another floor, putting the child at risk of being crushed or pinned and suffering serious injuries or death.
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response