Novel Laboratories is recalling approximately 5,700 boxes of Zolpidem Tartrate (Intermezzo) sublingual sleep tablets because the blister packaging is not child-resistant as required by federal law. The prescription medication is packaged in 30 unit-dose pouches per box and was sold at pharmacies including CVS, Costco, and Walgreens between March 2016 and May 2016. Because the packaging is easy for children to open, there is a serious risk of accidental ingestion. Consumers should stop using the tablets and contact the manufacturer for a full refund.
The drug packaging fails to meet child-resistant standards, allowing children to potentially open the blister packs and swallow the medication. This poses a significant risk of accidental poisoning for young children.
Consumers should immediately stop using the recalled tablets and contact Novel Laboratories for instructions to receive a full refund.
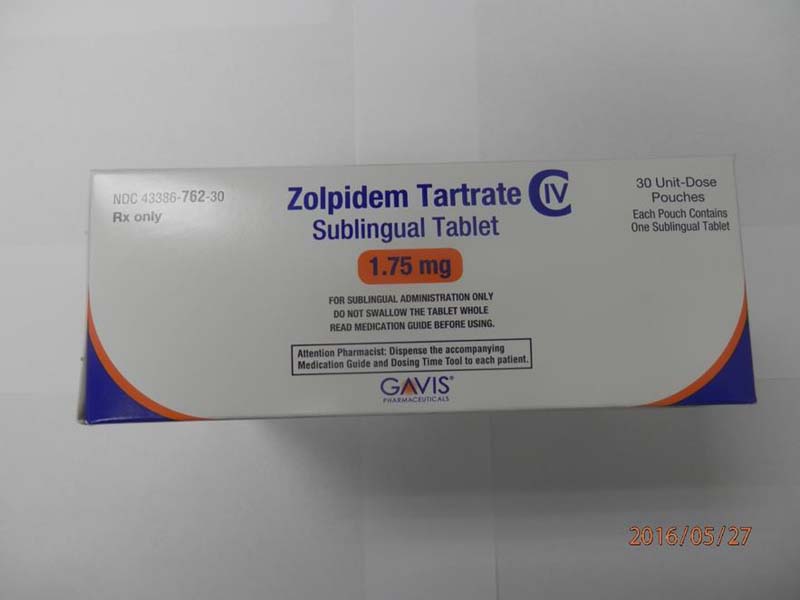
Novel Laboratories sleep tablets 1.75 mg box (front)

Novel Laboratories sleep tablets 1.75 mg envelope (front)

Novel Laboratories sleep tablets 1.75 mg envelope (back)
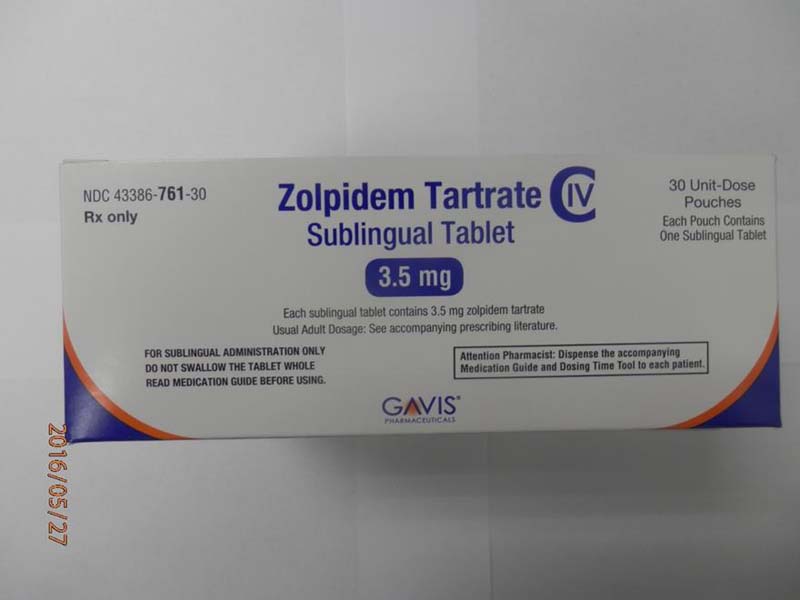
Novel Laboratories sleep tablets 3.5 mg box (front)
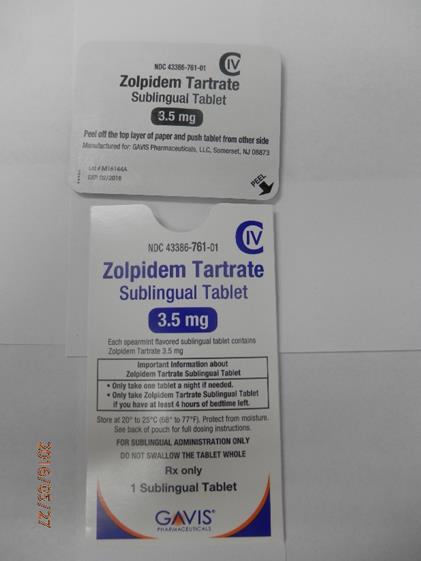
Novel Laboratories sleep tablets 3.5 mg envelope (front)

Novel Laboratories sleep tablets 3.5 mg envelope (back)
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response