Nanomaterials Discovery Corporation is recalling 500 bottles of Snowy Range Blue Alcohol Antiseptic 80% Topical Solution Hand Sanitizer (Alcohol 80% v/v). FDA analysis found the product to be contaminated with methanol, acetaldehyde, and acetal at levels exceeding safe limits. The recalled sanitizer is packaged in 4 fl. oz. (118 mL) blue bottles and was distributed nationwide by Reliable Products, LLC.
Methanol exposure can cause nausea, vomiting, headache, blurred vision, permanent blindness, seizures, coma, or permanent damage to the nervous system. Acetaldehyde and acetal are chemical contaminants that pose additional health risks if ingested or absorbed through the skin.
You have 2 options:
Distributed by Reliable Products, LLC, Cheyenne, WY 82003.

Snowy Range Blue, Hand Sanitizer, 4 fl. Oz. (front label)
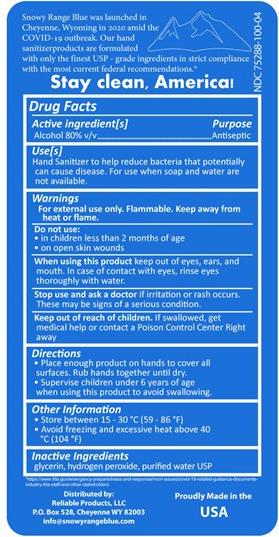
Snowy Range Blue, Hand Sanitizer, 4 fl. Oz. (back label)
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.