MOOR HERBS is recalling 147 units of Moor Herbs Healthy Beauty Angel Formula because the product was marketed as an infant formula without the required FDA pre-market safety notification. The 16-ounce plastic containers contain levels of iron, potassium, and sodium that are significantly higher than the maximum safe range for infants. Additionally, the formula lacks Vitamin D, which is essential for healthy bone development. The product was sold through the company's storefront in Detroit and via internet sales.
Infants consuming this product are at risk of iron overload and severe electrolyte imbalances due to high levels of sodium and potassium. Furthermore, the absence of Vitamin D can lead to rickets, a condition where a child's bones become soft and weak.
Return for refund

Product image, example of product code from the Soledad, CA facility
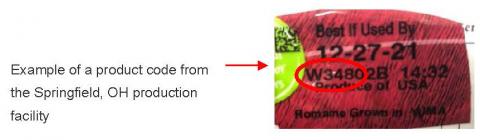
Product image, example of product code from the Springfield, OH production facility
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release (1) · FDA Press Release (2) · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.