Medique is recalling about 143,300 units of 31 different over-the-counter drug products under several brand names including Medi-First, Medique, and Otis Clapp because the packaging is not child-resistant as required by law. These medications, which include common pain relievers like aspirin, acetaminophen, and ibuprofen, as well as allergy and burn treatments, were sold exclusively on Amazon.com between June 2018 and June 2020. Consumers should immediately move these products to a secure location and contact Medique for disposal instructions and a full refund.
The packaging for these medications fails to meet child-resistant requirements, creating a significant risk of accidental poisoning if a young child is able to open the container and swallow the contents. While no injuries have been reported, many of these substances are highly toxic to children in small doses.
Consumers should immediately store the recalled products in a safe location out of reach of children and contact Medique for information on how to dispose of the product and receive a full refund. All known purchasers are being notified directly.
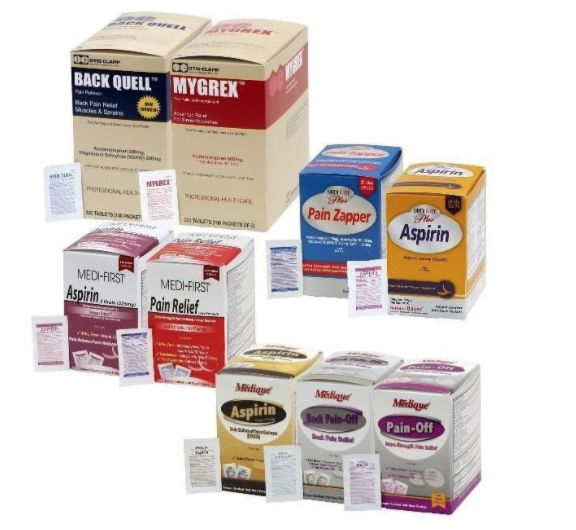
Recalled aspirin-containing products
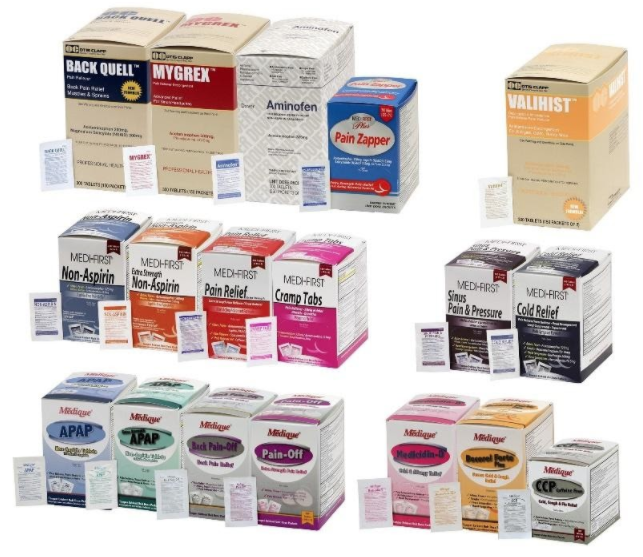
Recalled acetaminophen-containing products

Recalled diphenhydramine product
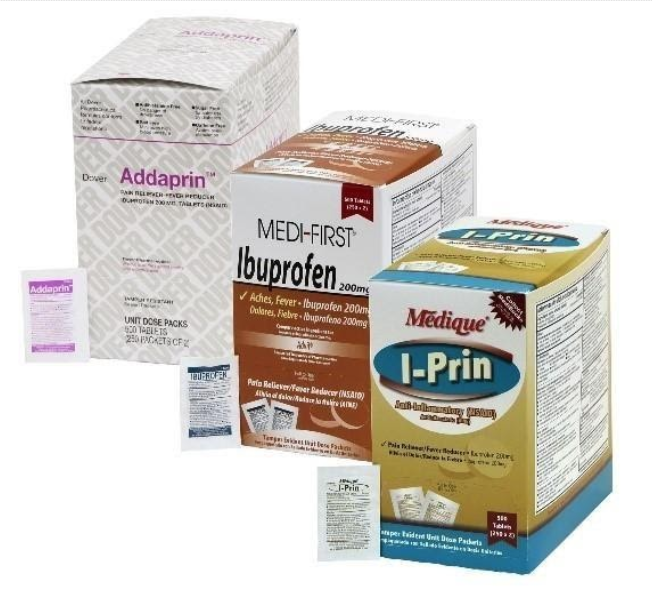
Recalled ibuprofen-containing products

Recalled lidocaine-containing products

Recalled loperamide product
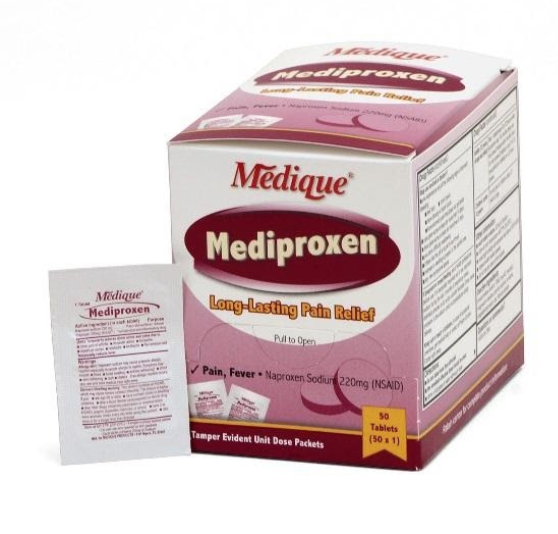
Recalled naproxen product
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response