Kinder Farms LLC is recalling 968,054 bottles of KinderMed KIDS' PAIN & FEVER and KinderMed INFANTS' PAIN & FEVER Acetaminophen (160 mg per 5 mL). These oral suspension medications in Organic Cherry Flavor were found to have failed quality specifications for impurities and degradation. No incidents or injuries have been reported to date.
The product failed to meet standards for impurities and degradation. Using medication that has broken down over time or contains excessive impurities may result in the medication being less effective or potentially causing unexpected side effects.
You have 2 options:
Distributed By: KinderFarms, Redondo Beach, CA 90277.
Distributed By: KinderFarms, Redondo Beach, CA 90277.
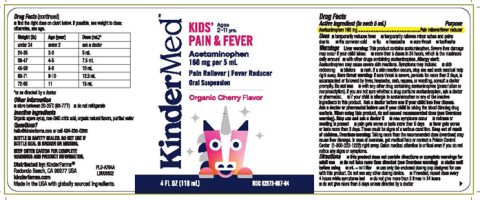
Image 1: “Bottle Label KinderMed Kids Pain & Fever Acetaminophen Organic Cherry Flavor, 4 fl. oz.”
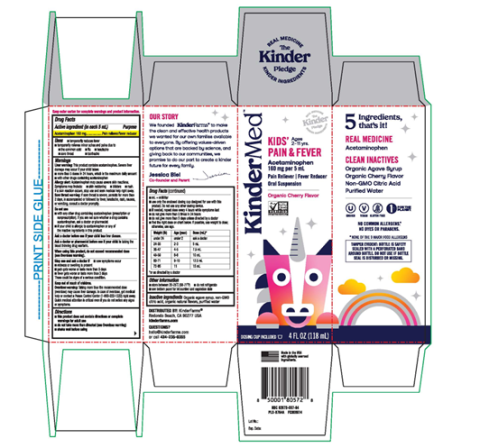
Image 2: “Carton Label KinderMed Kids Pain & Fever Acetaminophen Organic Cherry Flavor, 4 fl. oz.”
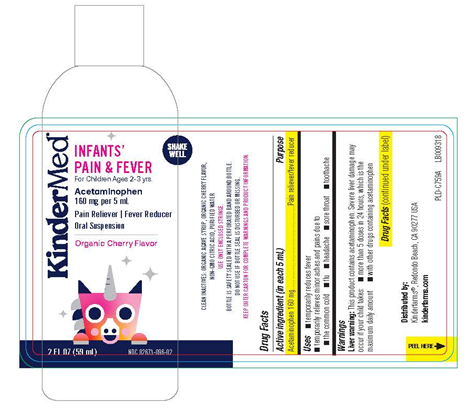
Image 3: “Bottle Label KinderMed Infants Pain & Fever Acetaminophen Organic Cherry Flavor, 2 fl. oz.”

Image 4: “Carton Label KinderMed Infants Pain & Fever Acetaminophen Organic Cherry Flavor, 2 fl. oz.”
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.