InfuTronix is recalling approximately 104,656 Nimbus Infusion Pump Systems, including the Nimbus Flex, PainPro, and Nimbus II models, because they are being removed from the market. These devices have multiple failure modes including battery failure, drug leakage, system errors, and incorrect flow rates. The manufacturer will stop supporting these devices after June 20, 2024, meaning they will no longer be available for use.
The pumps may fail to deliver medication accurately or stop working entirely due to battery or mechanical issues, which can lead to serious injury or death depending on the critical nature of the medication being infused.
Removal from market and discontinuation of support.
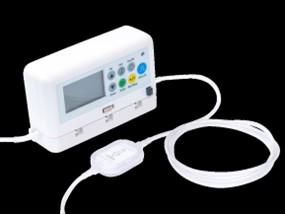
Nimbus Pump System Image
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.