Incredible Products, SA de CV is recalling Incredible Products Gelbac T Antibacterial Hand Gel (80% ethyl alcohol) because testing found the product contains methanol instead of the labeled ethyl alcohol. Methanol, or wood alcohol, is a toxic substance that is not an acceptable ingredient for hand sanitizers and can be dangerous when absorbed through the skin or swallowed. This recall affects both the 4.2-ounce (125 mL) bottles and the larger 33.8-ounce (1 L) bottles distributed in California and Georgia. Consumers who have these products should contact their healthcare provider or pharmacist for guidance and return any unused product to the place of purchase for a refund.
Substantial methanol exposure can result in nausea, vomiting, headache, blurred vision, permanent blindness, seizures, coma, permanent damage to the nervous system, or death. While all persons using these products on their hands are at risk, young children who accidentally ingest these products and adolescents or adults who drink these products as an alcohol (ethanol) substitute are at highest risk for methanol poisoning.
Drug product return and refund

Front label, GELBAC T ANTIBACTERIAL HANDGEL 125ml/4.2 oz.

Back label, GELBAC T ANTIBACTERIAL HANDGEL 125ml/4.2 oz.
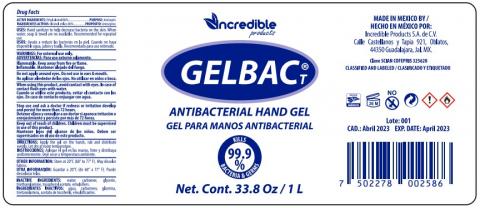
Label, GELBAC T ANTIBACTERIAL HANDGEL 1 liters /33.8 oz
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.