IBSA Pharma Inc. has voluntarily recalled multiple lots of Tirosint-Sol (levothyroxine sodium) Oral Solution because the medication may be subpotent. This means the solution might not contain the full amount of active medicine required to treat thyroid conditions effectively. The recall affects 15 different dosage strengths, each packaged in boxes containing six pouches of five ampules. No injuries or adverse events related to this issue have been reported to date.
Subpotent medication can lead to inadequate treatment of hypothyroidism (underactive thyroid), potentially resulting in symptoms such as fatigue, weight gain, and cold intolerance. There is a risk that patients may not receive their intended dose, leading to poor control of their thyroid levels.
You have 2 options:
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.
Rx Only; Manufactured for IBSA Pharma Inc. by IBSA Institut Biochimique SA.

1. “TIROSINT-SOL 13 mcg/mL 30 units carton-box, NDC 71858-0105-5”

2. “TIROSINT-SOL 25 mcg/mL 30 units carton-box, NDC 71858-0110-5”

3. “TIROSINT-SOL 37.5 mcg/mL 30 units carton-box, NDC 71858-0112-5”

4. “TIROSINT-SOL 44 mcg/mL 30 units carton-box, NDC 71858-0113-5”

5. “TIROSINT-SOL 50 mcg/mL 30 units carton-box, NDC 71858-0115-5”

6. “TIROSINT-SOL 62.5 mcg/mL 30 units carton-box, NDC 71858-0117-5”

7. “TIROSINT-SOL 75 mcg/mL 30 units carton-box, NDC 71858-0120-5”

8. “TIROSINT-SOL 88 mcg/mL 30 units carton-box, NDC 71858-0125-5”

9. “TIROSINT-SOL 100 mcg/mL 30 units carton-box, NDC 71858-0130-5”
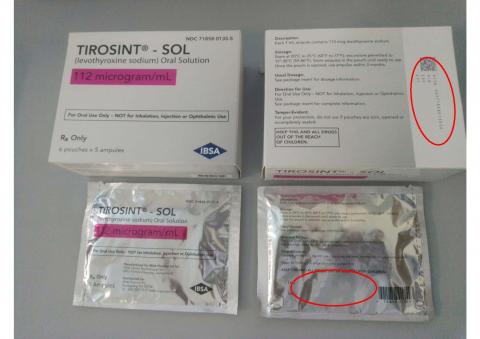
10. “TIROSINT-SOL 112 mcg/mL 30 units carton-box, NDC 71858-0135-5”

11. “TIROSINT-SOL 125 mcg/mL 30 units carton-box, NDC 71858-0140-5”

12. “TIROSINT-SOL 137 mcg/mL 30 units carton-box, NDC 71858-0145-5”

13. “TIROSINT-SOL 150 mcg/mL 30 units carton-box, NDC 71858-0150-5”

14. “TIROSINT-SOL 175 mcg/mL 30 units carton-box, NDC 71858-0155-5”

15. “TIROSINT-SOL 200 mcg/mL 30 units carton-box, NDC 71858-0160-5”
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.