Green Lumber Holdings, LLC is recalling Green Lumber Natural Fuel For Men capsules because FDA testing found the product contains tadalafil, an undeclared drug ingredient. Tadalafil is the active ingredient in Cialis, a prescription drug used for erectile dysfunction, and its presence makes this product an unapproved drug. Consumers should contact their healthcare provider or pharmacist if they have concerns, and return the product for a refund or throw it away.
The undeclared tadalafil can interact with nitrates found in some prescription drugs (such as nitroglycerin), potentially lowering blood pressure to dangerous levels. This poses a significant risk to consumers with diabetes, high blood pressure, high cholesterol, or heart disease who often take nitrates.
You have 2 options:
Recall #: D-0304-2026; distributed via GreenLumber.com
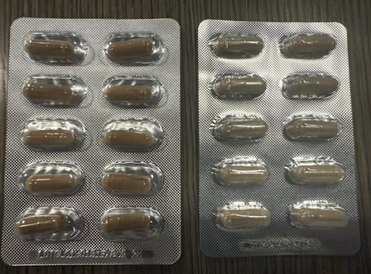
Authentic Capsule Package on Left, Counterfeit Package on Left

Front Panel for Green Lumber Natural Fuel For Men, 10 capsules

Back Panel for Green Lumber Natural Fuel For Men, 10 capsules
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release (1) · FDA Press Release (2) · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.