Golden State Medical Supply Inc. is recalling 2,584 bottles of Atenolol (25 mg) tablets because some bottles were mistakenly filled with Clopidogrel (75 mg) tablets. Atenolol is used to treat high blood pressure and chest pain, while Clopidogrel is a blood thinner. The recalled products are 1,000-count bottles that were distributed nationwide. Patients may notice that instead of the expected white tablets, the bottle contains pink tablets.
Patients who unknowingly take Clopidogrel instead of their prescribed Atenolol will miss their necessary blood pressure medication and receive a blood thinner they were not intended to have. This can cause uncontrolled blood pressure or heart conditions and significantly increases the risk of dangerous, unintended bleeding.
You have 2 options:
Recalled bottles contain Clopidogrel 75mg tablets instead of the labeled Atenolol 25mg tablets.

Product labeling, Clopidogrel Tablets, USP 75mg 1,000 count bottles, NDC 51407-032-10

Product image, Clopidogrel Tablets
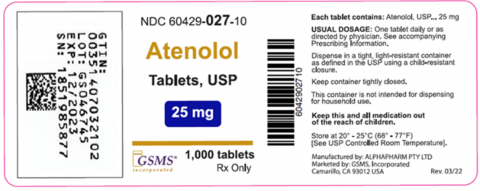
Product labeling, Atenolol Tablets, USP 25mg 1,000 count bottles NDC 60429-027-10
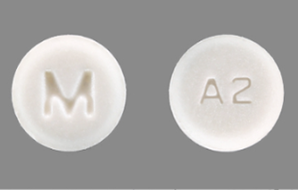
Product image, Atenolol Tablets
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.