DermaRite Industries, LLC is recalling 24,246 units of various hand hygiene and skin care products, including DermaSarra, DermaKleen, KleenFoam, and PeriGiene. These products, which include antiseptic soaps and analgesic lotions, were found to be contaminated with the bacteria Burkholderia cepacia. Consumers who have these products should stop using them and contact their healthcare provider if they have concerns, as the bacterial contamination poses a risk of infection.
The products are contaminated with Burkholderia cepacia bacteria, which can cause serious infections, particularly in individuals with weakened immune systems or underlying lung conditions. Use of contaminated topical products or soaps can lead to skin or systemic infections.
You have 2 options:
Recall #: D-0611-2025; Quantity: 249 bottles
Recall #: D-0610-2025; Quantity: 11,321 bag in boxes
Recall #: D-0612-2025; Quantity: 6,973 cartridges
Recall #: D-0613-2025; Quantity: 5,703 bottles

DermaKleen, Dermasarra, Kleenfoam, and Perigiene Products Table

DermaKleen, Dermasarra, Kleenfoam, and Perigiene Products Table

Label, PeriGiene Healthcare Antiseptic

Label, DermaKleen Health Care Antiseptic Lotion Soap with Vitamin E, 1000mL

Label, DermaKleen Health Care Antiseptic Lotion Soap with Vitamin E, 800mL

Label, KleenFoam Antimicrobial Foam Soap

Label, DermaSarra External Analgesic

Recalled Product List page 1
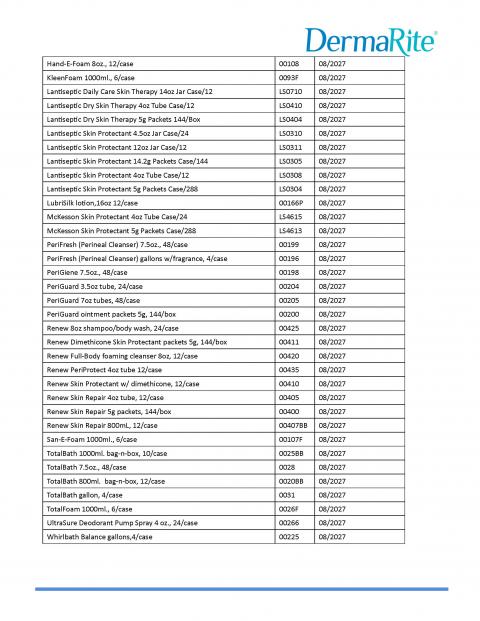
Recalled Product List page 2

“DermaRite DermaCerin”

“DermaRite DermaFungal”

“DermaRite DermaKleen”

“DermaRite DermaMed”

“DermaRite DermaSarra”

“DermaRite GelRite”

“DermaRite Hand-E-Foam”

“DermaRite KleenFoam”
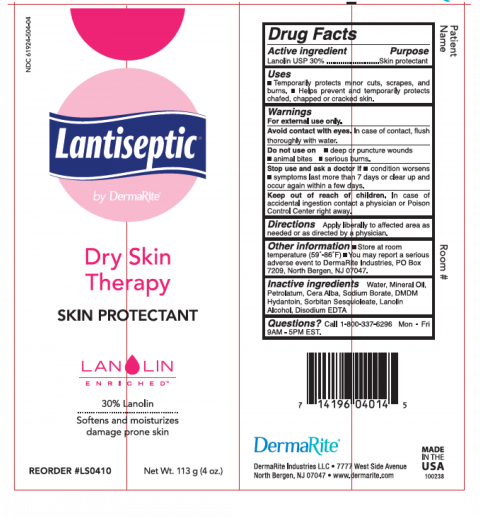
“DermaRite Lantiseptic”

“DermaRite PeriGiene”

“DermaRite PeriGuard”

“DermaRite Renew Dimethicone Skin Protectant”

“DermaRite Renew Periprotect Skin Protectant”

“DermaRite Renew Skin Repair Cream”

“DermaRite UltraSure Anti-Perspirant & Deodorant”

“DermaRite 4-N-1 Skin Protectant”
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release (1) · FDA Press Release (2) · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.