DermaRite Industries, LLC is recalling approximately 766,166 units of various skin protectants, antifungal creams, hand sanitizers, and soaps, including brands like Lantiseptic, GelRite, and PeriGuard. These products were manufactured at a facility where other product lots were found to be contaminated, posing a risk of infection. Affected items were distributed nationwide in the United States and include numerous sizes ranging from 5g packets to 1000mL cartridges.
The products were manufactured under conditions that deviated from safe manufacturing practices (CGMP), specifically at a facility where contamination was confirmed in other items. This creates a risk that these skin-contact products may contain harmful microorganisms, which could lead to infections, particularly in individuals with compromised immune systems or broken skin.
You have 2 options:
Quantity: 8,378 containers
Quantity: 3,178 bottles
Quantity: 1,480 packets
Quantity: 7,736 tubes
Quantity: 2,673 tubes
Quantity: 2,389 bottles
Quantity: 115,994 containers
Quantity: 576 containers
Quantity: 15,267 cartridges
Quantity: 57,837 units
Quantity: 5,876 units
Quantity: 16,006 units
Quantity: 381,619 units
Quantity: 37,272 tubes
Quantity: 57,041 units
Quantity: 15,465 cartridges
Quantity: 37,379 spray bottles

Recalled Product List page 1
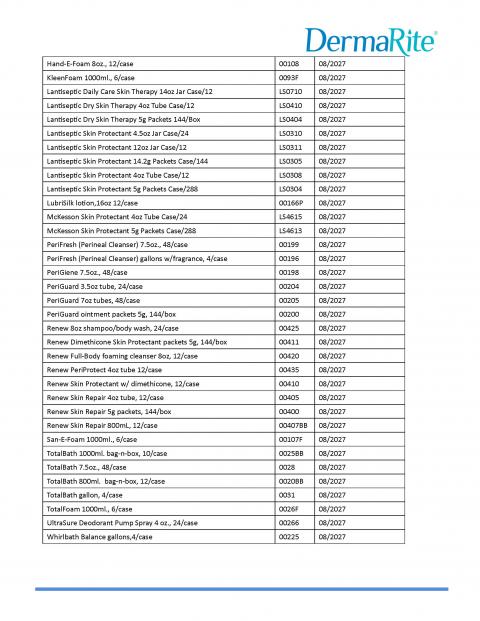
Recalled Product List page 2

“DermaRite DermaCerin”

“DermaRite DermaFungal”

“DermaRite DermaKleen”

“DermaRite DermaMed”

“DermaRite DermaSarra”

“DermaRite GelRite”

“DermaRite Hand-E-Foam”

“DermaRite KleenFoam”
“DermaRite Lantiseptic”

“DermaRite PeriGiene”

“DermaRite PeriGuard”

“DermaRite Renew Dimethicone Skin Protectant”

“DermaRite Renew Periprotect Skin Protectant”

“DermaRite Renew Skin Repair Cream”

“DermaRite UltraSure Anti-Perspirant & Deodorant”

“DermaRite 4-N-1 Skin Protectant”
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.