Covidien is recalling approximately 5,709 McGRATH MAC Video Laryngoscopes because these specific units were stolen after failing quality tests and being designated for destruction. These defective devices were subsequently sold and distributed illegally, and they may lack proper packaging, labeling, or serial numbers. Using these failed devices in a medical setting can result in a failed laryngoscopy or intubation, which are critical procedures for maintaining a patient's airway.
The use of these defective laryngoscopes may lead to an inability to see the vocal cords or successfully place a breathing tube. This can result in respiratory failure, low oxygen levels, or other life-threatening complications during emergency or surgical procedures.
Quarantine and identify affected units.

Image 1: “Covidien McGRATH MAC Video Laryngoscope device”
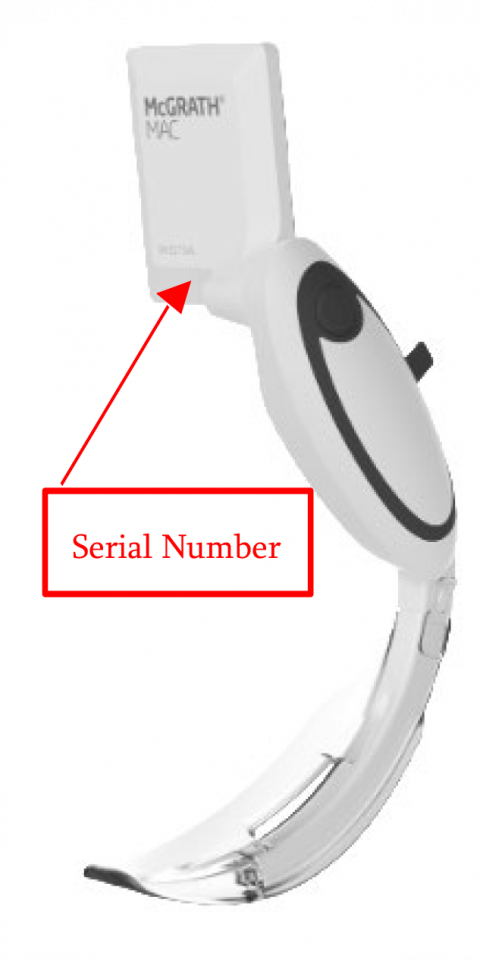
Image 2: “McGRATH MAC Video Laryngoscope device”

Image 3: “Covidien McGRATH MAC Video Laryngoscope device container”

Image 4: “McGRATH MAC Video Laryngoscope device container”
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.