Bryant Ranch Prepack, Inc. is recalling two lots of Morphine Sulfate extended-release tablets (morphine sulfate) due to a labeling error. Bottles labeled as containing 60 mg tablets may actually contain 30 mg tablets, and bottles labeled as 30 mg may contain 60 mg tablets. This error occurred during the packaging process and affects 100-count bottles of both strengths. While no injuries have been reported, taking an incorrect dose of this medication poses a high risk to patient safety.
Consumers taking the 60 mg dose could accidentally receive the 30 mg strength, leading to inadequate pain control and potential withdrawal. Conversely, patients prescribed the 30 mg dose who receive the 60 mg strength are at risk of a life-threatening overdose.
You have 2 options:
Manufactured by Ohm Laboratories Inc.; Relabeled by Bryant Ranch Prepack, Inc.
Manufactured by Ohm Laboratories Inc.; Relabeled by Bryant Ranch Prepack, Inc.

Product image, 30mg tablets round, purple-colored

Product image, label 30mg
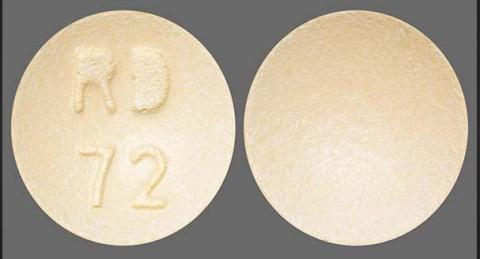
Product image, 60mg tablets round, light orange-colored

Product image, label 60mg
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.