Kroger is recalling approximately 25,660 bottles of Kroger brand Acetaminophen Arthritis Pain Extended-Release Tablets (650 mg). The product's packaging is not child-resistant as required by the Poison Prevention Packaging Act, which poses a serious poisoning risk if children access and swallow the medication. No incidents or injuries have been reported at this time. The affected bottles were sold in various Kroger-affiliated stores nationwide between December 2021 and March 2022.
The packaging fails to meet child-resistant standards, allowing young children to potentially open the bottle and ingest the tablets. Acetaminophen poisoning can cause severe liver damage or death in young children.
Consumers should immediately store the recalled product in a safe location out of reach and sight of children. Contact Kroger for information on how to properly dispose of the product and receive a full refund.
Red white and yellow label; red continuous thread gear closure.
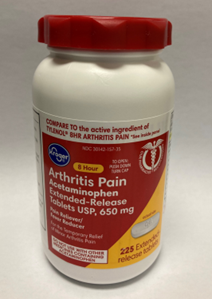
Recalled Kroger Arthritis Pain Acetaminophen 650mg, 225 count bottle

The UPC code, lot number and expiration date are printed on the label on the back of the bottle
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: CPSC Notice · Raw API Response