Apotex Corp. is voluntarily recalling 67,056 bottles of Brimonidine Tartrate Ophthalmic Solution 0.15%, a prescription eye drop (Brimonidine Tartrate) used to lower eye pressure in patients with glaucoma or ocular hypertension. The recall was issued because cracks were discovered in the bottle caps, which could allow contaminants into the bottle and compromise the sterility of the medicine. No injuries or adverse events related to this issue have been reported to date.
A cracked bottle cap could compromise the sterile seal of the eye drops, allowing bacteria or other contaminants to enter the solution. Using non-sterile eye medication poses a serious risk of eye infections and other related health complications.
You have 2 options:
Manufactured by Apotex Inc. Toronto, Ontario Canada.
Manufactured by Apotex Inc. Toronto, Ontario Canada.
Manufactured by Apotex Inc. Toronto, Ontario Canada.
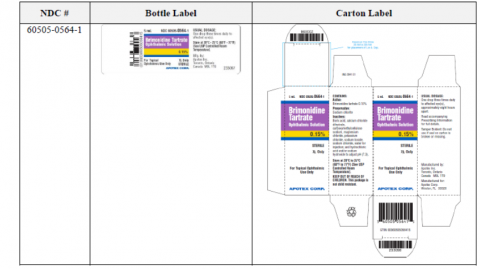
Image 1 – Labeling, Brimonidine Tartrate Ophthalmic Solution, 0.15%, Bottle Label
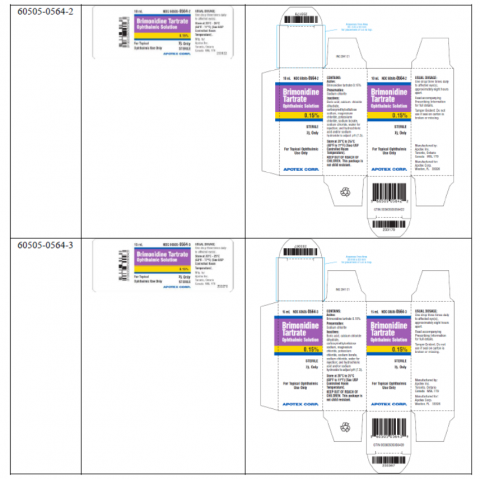
Image 2 – Labeling, Brimonidine Tartrate Ophthalmic Solution, 0.15%, Carton Label
If you or a family member were harmed by this recalled product, you may have legal rights. Consider consulting a consumer protection attorney to understand your options for compensation.
This is general information, not legal advice. Go Backs is not a law firm and does not provide legal services.
AI-Enhanced Content: The summary, action steps, and risk assessment on this page were generated by AI from official government recall data to improve readability. This is not legal or medical advice. Always refer to the official agency sources below for authoritative information.
Sources: FDA iRES · FDA Press Release · Raw API Response
openFDA Disclaimer: This recall information is sourced from the openFDA API. Do not rely on openFDA to make decisions regarding medical care. While we make every effort to ensure that data is accurate, you should assume all results are unvalidated. Refer to the openFDA terms of service for more information.